 
Subsequently, accelerator-produced 67Ga (t 1/2 = 3.3 d) was investigated for clinical use, and determined to be an effective tumor and abscess locating agent, with annual usage reaching nearly 250,000 patients by 1977 (Hayes 1978). A primary factor contributing to the negative diagnostic results was the poor detection equipment available at the time, while any further attempts exploit 72Ga for therapy would have been limited by the high energy and intensity beta particle and gamma ray emissions depositing excess radiation dose in healthy tissue surrounding the tumor sites. Early clinical trials using reactor-produced 72Ga (t 1/2 = 14.1 h) for therapy and diagnostic evaluation of malignant bone lesions were ineffective, with investigation largely stopping by 1952 due to unsatisfactory patient benefits (Hayes 1978). Radiogallium isotopes have been extensively investigated, starting when gallium was first observed to accumulate at osteogenic activity in the late 1940s (Hayes 1978). The rise and increasingly widespread clinical use of positron emission tomography (PET) imaging with gallium-68 ( 68Ga) radiopharmaceuticals motivates providing guidance on aspects of 68Ga radiopharmaceutical production to aid the community in achieving consistent quality and reliable yields. Finally, an outlook on 68Ga radiopharmaceuticals is presented to highlight potential challenges and opportunities facing the community. This review outlines important aspects of 68Ga radiopharmacy, including 68Ga production routes using a 68Ge/ 68Ga generator or medical cyclotron, standardized 68Ga radiolabeling methods, quality control procedures for clinical 68Ga radiopharmaceuticals, and suggested best practices for centers with established or upcoming 68Ga radiopharmaceutical production. This progress motivates the development of a set of common guidelines and standards for the 68Ga radiopharmaceutical community, and recommendations for centers interested in establishing 68Ga radiopharmaceutical production. The short half-life, favourable nuclear decay properties, ease of radiolabeling, and convenient availability through germanium-68 ( 68Ge) generators and cyclotron production routes strongly positions 68Ga for continued growth in clinical deployment. These 68Ga radiopharmaceuticals include agents such as Ga-macroaggregated albumin for myocardial perfusion evaluation, Ga-PLED for assessing renal function, Ga- t-butyl-HBED for assessing liver function, and Ga-PSMA for tumor imaging. In precision medicine, PET applications of 68Ga are widespread, with 68Ga radiolabeled to a variety of radiotracers that evaluate perfusion and organ function, and target specific biomarkers found on tumor lesions such as prostate-specific membrane antigen, somatostatin, fibroblast activation protein, bombesin, and melanocortin. Responses due are to be submitted by e-mail only to Name of firm, Socio-Economic category of the firm is needed, capable (SDVOSB & VOSB) vendors should supply CVE Certification letter from Veterans Information Pages (VIP), along with complete address, phone & fax number of contacts.The radiometal gallium-68 ( 68Ga) is increasingly used in diagnostic positron emission tomography (PET), with 68Ga-labeled radiopharmaceuticals developed as potential higher-resolution imaging alternatives to traditional 99mTc agents. The applicable North American Classification System (NAICS) Code for this requirement is 325412 (Pharmaceutical Preparation Manufacturing). Please be advised that the Government will not pay for any information submitted under this announcement. THIS IS NOT A SOLICITATION AND DOES NOT OBLIGATE THE GOVERNMENT TO ISSUE A SOLICITATION. The Department of Veteran s Affairs is seeking small business manufacturer or distributors of small business manufacturers, of functionally Brand-Name Only to all the following: NetSpot - Ga-68 Dotatate Item # Description/Part Number* Qty 1 VAMC, New York Harbor, Netspot (Ga-68 Netspot) 6 2 VAMC, East Orange, Netspot (Ga-68 Netspot) 12 3 VAMC, Bronx, Netspot (Ga-68 Netspot) 12 4 VAMC, Norhtport, Netspot (Ga-68 Netspot) 5 RESPONSE DATE (DEADLINE): at 11:00 AM. 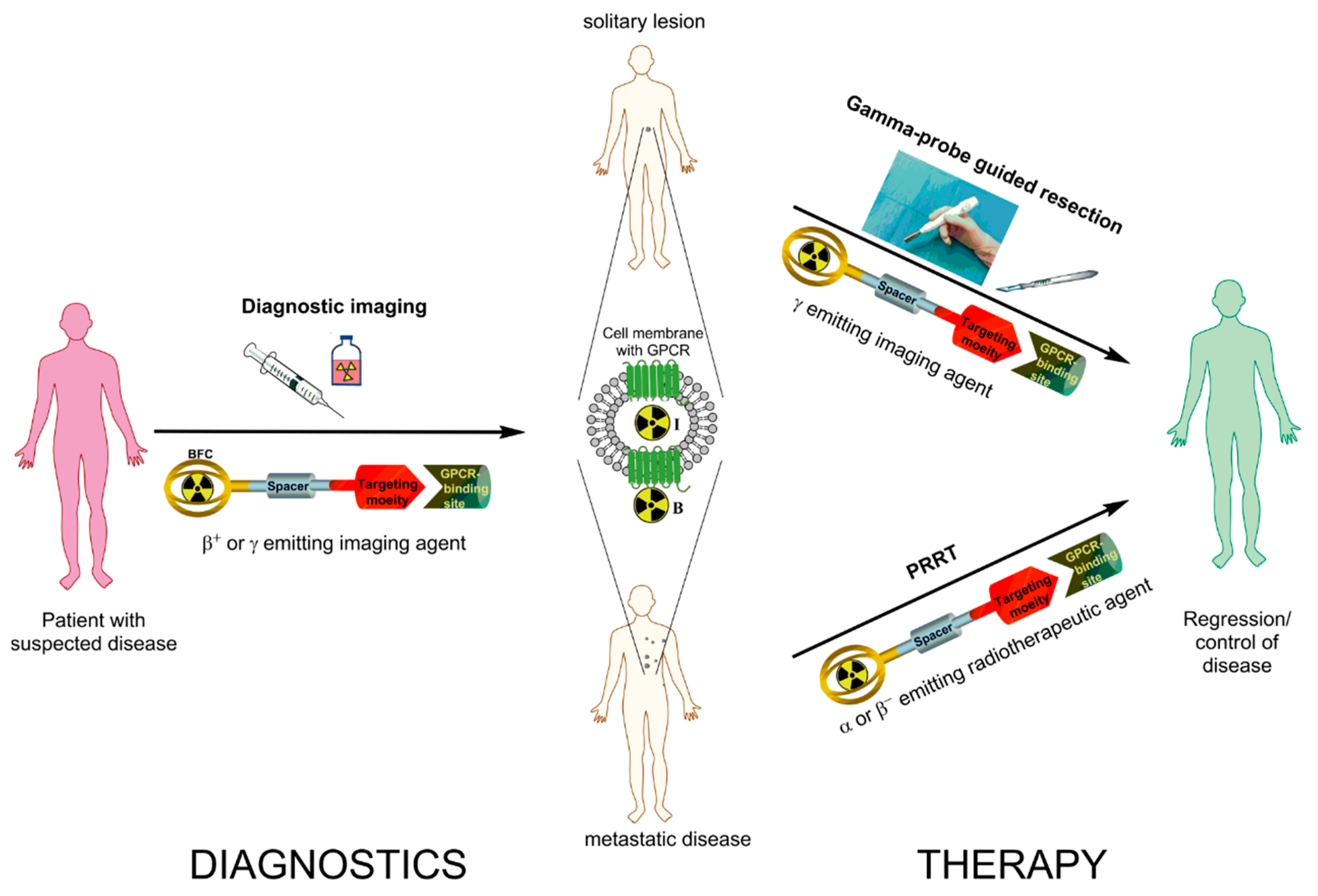
The purpose of this Sources Sought notice is to determine interest and capability of potential qualifications relative to the North American Industry Classification Code (NAICS) 325412. This notice is intended strictly for market research. 
This Sources Sought Notice is for informational and planning purposes only and shall not be construed as a solicitation or as an obligation or commitment by the Government at this time. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
